Tag: Drug Approvals
FDA Gives Moderna COVID-19 Vaccine Full Approval
Vaccine, which has been available for the age group under emergency use authorization, will be marketed as Spikevax
FDA Approves Rinvoq for Treatment of Atopic Dermatitis
Approval indicated for those 12 years and older with atopic dermatitis that has not responded to previous treatment
Most COVID-19-Related EUAs Not Supported by High-Quality Evidence
Most EUAs were for diagnostic products; most supported by comparisons with previously authorized assays, analytical in vitro studies
FDA Approves New Drug to Help Control Severe Asthma
Tezspire is unique in that it only targets thymic stromal lymphopoietin, the molecule involved in airway inflammation
FDA Gives OK to Merck Antiviral At-Home COVID-19 Pill
Molnupiravir cut the risk for hospitalization and death in high-risk patients by 30 percent
FDA Approves Pfizer Pill as First At-Home COVID-19 Treatment
Recent trial showed Paxlovid cut the odds for hospitalization and death by nearly 90 percent in high-risk people
FDA Approves First-in-Class Treatment for Myasthenia Gravis
Vyvgart is the first neonatal Fc receptor blocker to be approved to treat generalized myasthenia gravis in patients testing positive for AChR antibody
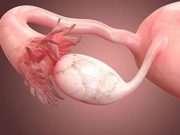
FDA Approves Imaging Drug That Can Help Surgeons Spot Ovarian Cancers
Drug is designed to improve surgeon's ability to detect ovarian cancer while operating on a patient
FDA Approves First Interchangeable Biosimilar for Inflammatory Diseases
Cyltezo is first interchangeable monoclonal antibody for RA, psoriatic arthritis, ankylosing spondylitis, Crohn disease, ulcerative colitis, chronic plaque psoriasis
First Biosimilar Approved for Macular Degeneration, Other Eye Conditions
Biosimilar to Lucentis approved to treat neovascular age-related macular degeneration, macular edema, myopic choroidal neovascularization