Phase 3 trials show significant reduction in total lesion count versus Oracea and placebo
By Lori Solomon HealthDay Reporter
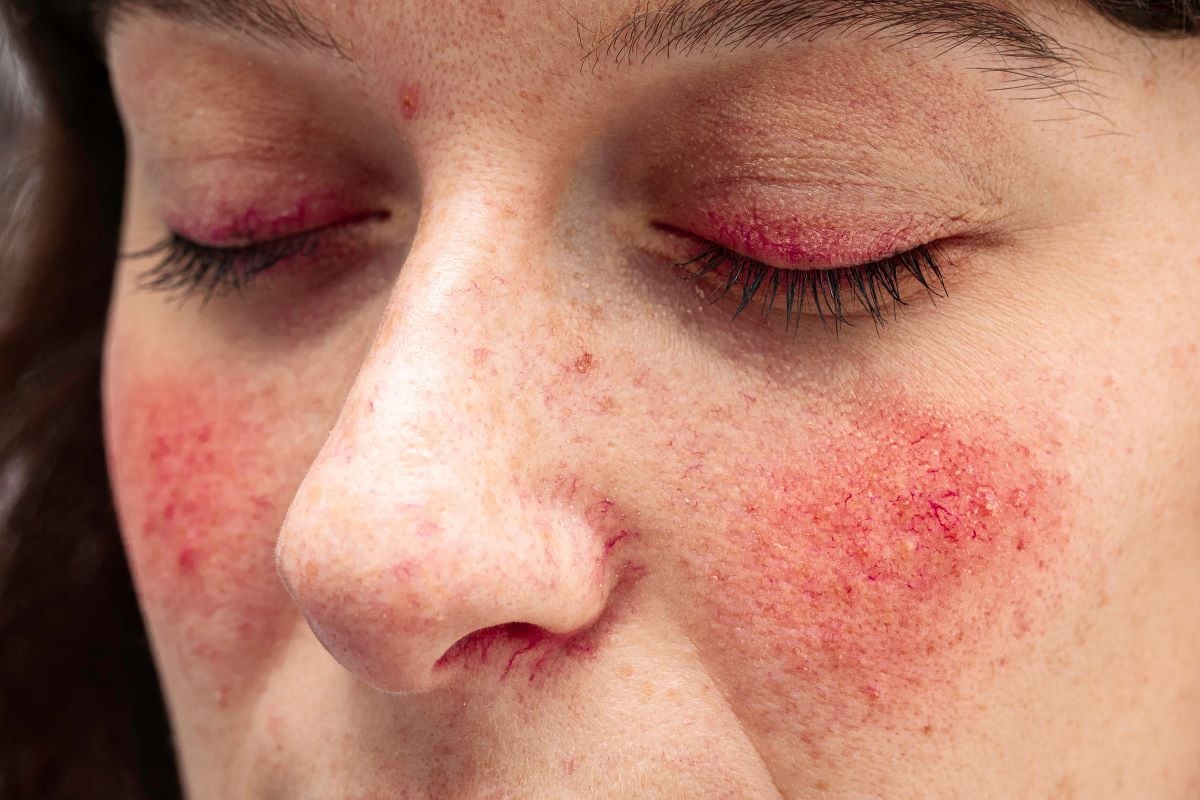
TUESDAY, Nov. 12, 2024 (HealthDay News) — The U.S. Food and Drug Administration has approved Emrosi (minocycline hydrochloride) for the treatment of inflammatory rosacea in adults.
The extended-release capsules (40 mg) will be available late in the first quarter or early in the second quarter of 2025.
The approval was based on positive results from two 16-week phase 3 clinical trials, during which Emrosi met all coprimary and secondary end points and showed significant superiority over both the current standard-of-care treatment (Oracea) and placebo for Investigator’s Global Assessment treatment success as well as reduction in total inflammatory lesion count. Additionally, participants experienced no significant safety issues.
“Rosacea is a difficult to treat skin condition, and based on the favorable results from our phase 3 clinical trials, Emrosi has potential to become the best-in-class oral medication to treat the condition,” Claude Maraoui, the cofounder, president, and CEO of Journey Medical, said in a statement. “Our seasoned dermatology-focused sales force is now preparing for a successful launch and to establish Emrosi as a new standard of care in the treatment of rosacea.”
The approval of Emrosi was granted to Journey Medical.
Copyright © 2024 HealthDay. All rights reserved.